
With larger ketones, we must use a locant number to indicate the position of the carbonyl group just before the suffix, as we did with alkenes and alkynes: 
The common name for propanone is acetone. When naming a ketone, we take the name of the parent hydrocarbon and change the suffix to – one: The smallest ketone has three C atoms in it. Despite the fact that aldehydes and ketones have the same carbonyl group, they have different chemical and physical properties and are properly grouped as two different types of compounds. This type of molecule is called a ketone. The main thing to note about aldehydes is that the carbonyl group is at the end of a carbon chain.Ī carbonyl group in the middle of a carbon chain implies that both remaining bonds of the carbonyl group are made to C atoms. Methanal has a common name with which you may be familiar: formaldehyde. (Do not confuse – al with – ol, which is the suffix used for alcohols.) So we have: The parent name of the hydrocarbon is used, but the suffix – al is appended. When naming aldehydes, the main chain of C atoms must include the carbon in the carbonyl group, which is numbered as position 1 in the carbon chain. 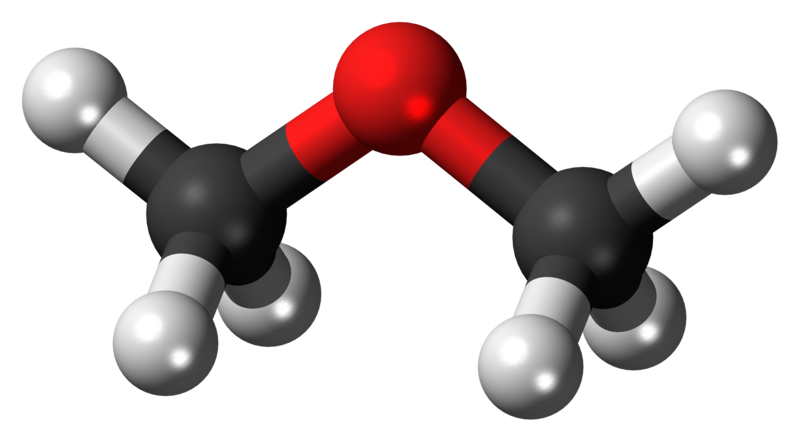
If one bond of the carbonyl group is made to a hydrogen atom, then the molecule is further classified as an aldehyde. In this diagram, the R group represents any hydrocarbon chain: A carbonyl group is formed when an O atom and a C atom are joined by a double bond. There are other functional groups that contain oxygen atoms. Use proper naming conventions for aldehyde, ketone, carboxylic acid, and ester- and ether-containing molecules.Identify the aldehyde, ketone, acid, ester, and ether functional groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
